You pull a favorite bracelet from your drawer, and instead of the rich luster you remember, there’s a dull, discolored film sitting on the surface. If you’ve ever wondered why does jewelry tarnish, the answer comes down to chemistry, specifically, how certain metals react with moisture, oxygen, and sulfur compounds in the air. It’s not a sign of poor quality. It’s a natural process that affects even premium materials, from sterling silver to iron-based minerals like pyrite.
At Natural Pyrite UAE, we craft pyrite bracelets and decorative pieces meant to be worn and displayed daily. That means we think about tarnish constantly, how it forms, what accelerates it, and how to protect against it. The UAE’s humid climate and coastal air create conditions that can speed up oxidation on many types of jewelry, making this topic especially relevant for anyone building a collection here in Dubai, Abu Dhabi, or anywhere across the Emirates.
This article breaks down the specific chemical reactions behind tarnish, identifies which metals are most vulnerable, and gives you straightforward prevention methods that actually work. Whether you’re protecting a pyrite bracelet, a silver cuff, or a copper-accented statement piece, you’ll walk away knowing exactly how to keep your jewelry looking the way it should.
Why jewelry tarnishes in the first place
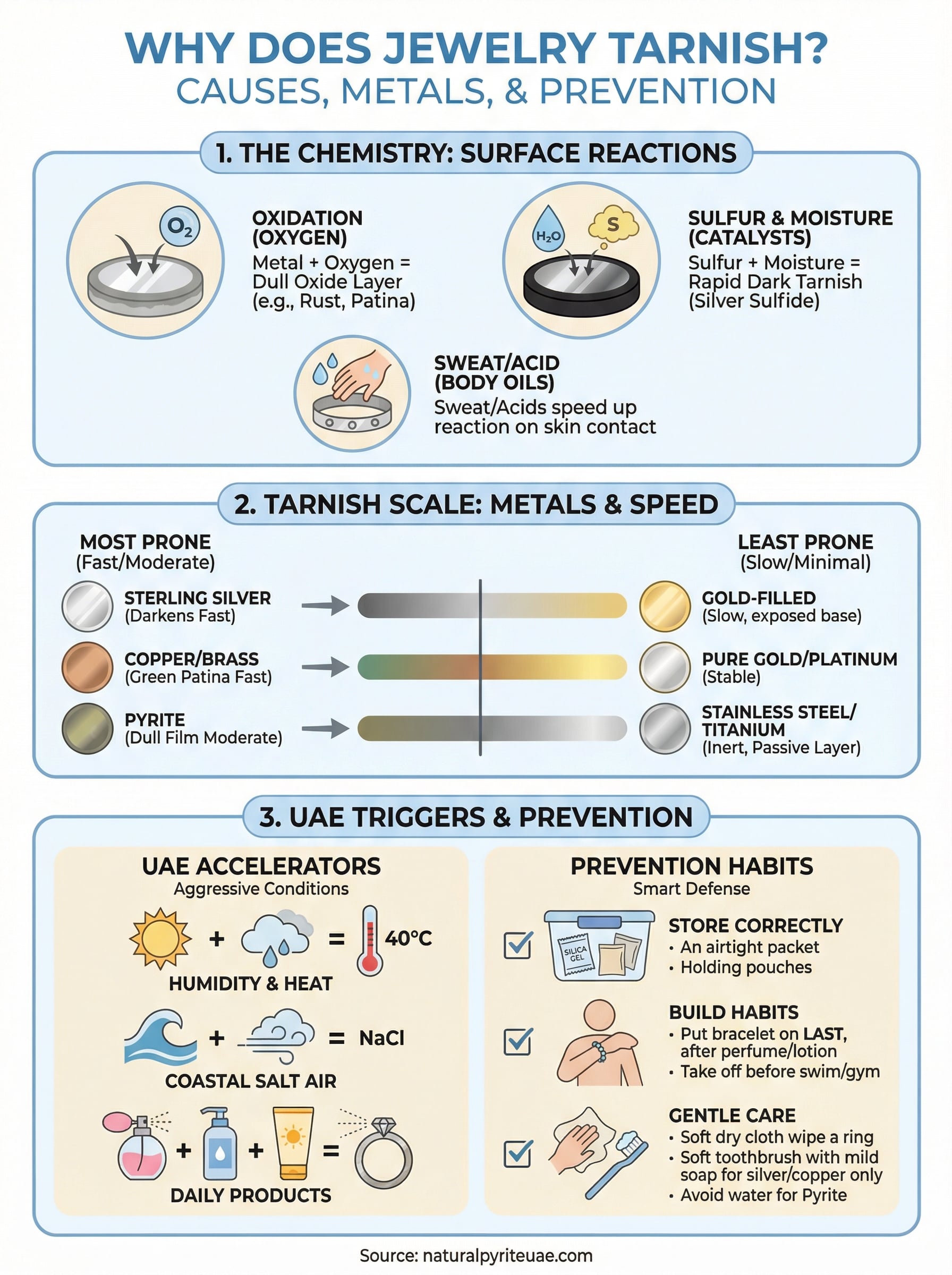
Tarnish isn’t damage in the traditional sense. It’s a surface-level chemical reaction where metal atoms bond with elements in the surrounding environment, most commonly oxygen, sulfur, and water vapor. The result is a thin layer of metal compound, such as silver sulfide or iron oxide, that sits on top of the original metal and changes how light reflects off it. Understanding this reaction is the first step in answering why does jewelry tarnish and knowing what to do about it.
The role of oxidation
When metal surfaces come into contact with oxygen in the air, they begin to oxidize. Oxidation is the process where metal atoms lose electrons to oxygen molecules, forming a new compound on the surface. You’ve seen this in everyday life: the rust on iron, the green patina on copper statues, the dark film on old silver rings. Each of these is an oxidation reaction specific to that metal’s chemistry. Some metals, like platinum and pure gold, have stable atomic structures that resist giving up electrons, so they oxidize far more slowly under normal conditions.
The thinner and more porous a metal’s surface finish, the faster oxidation takes hold, which is why polished, high-density metals hold their appearance longer.
How sulfur and moisture speed up the reaction
Oxygen alone doesn’t tell the full story. Sulfur compounds in the environment are a major driver of tarnish, especially for silver and copper-based alloys. These compounds come from everyday sources like rubber bands, wool fabric, certain foods, and polluted urban air. When sulfur bonds with silver, it produces silver sulfide, which is the dark, near-black layer you often see on aged silverware or old rings. Moisture acts as a catalyst in this process, giving the reaction a medium to travel through and significantly shortening the time it takes for visible tarnish to appear.
Sweat also plays a direct role. The salt and amino acids in human perspiration react with metal surfaces every time you wear a piece against your skin. This is why jewelry worn frequently in warm climates tends to tarnish faster than pieces kept in storage. Your body chemistry matters as well, since some people’s skin is naturally more acidic, which accelerates surface reactions on metals like copper, brass, and iron-containing minerals such as pyrite.
Metals that tarnish most and least
Not all metals react to the environment at the same rate. The atomic structure of a metal determines how readily it bonds with oxygen, sulfur, and moisture, which is the core of why does jewelry tarnish at different speeds depending on what it’s made from. Knowing which materials are more reactive helps you make smarter choices about storage, cleaning frequency, and daily wear.
Metals most prone to tarnish
Silver, copper, and brass are the most reactive metals in everyday jewelry. Sterling silver (92.5% silver) darkens relatively quickly because silver has a strong affinity for sulfur compounds. Copper and brass turn greenish as copper oxide and copper carbonate form on the surface. Pyrite, an iron sulfide mineral, also oxidizes when exposed to humidity, producing a dull surface film if not properly sealed or maintained.

The more alloy metals a piece contains, the more reactive sites exist on its surface, which means faster and more visible tarnish.
| Metal | Tarnish Speed | Primary Cause |
|---|---|---|
| Sterling silver | Fast | Sulfur compounds |
| Copper / brass | Moderate to fast | Oxidation |
| Pyrite | Moderate | Humidity and oxygen |
| Gold-filled | Slow | Thin base metal exposure |
| Pure gold / platinum | Minimal | Stable atomic structure |
Metals that resist tarnish
Pure gold and platinum sit at the opposite end of the spectrum. Both metals have highly stable atomic structures, meaning they don’t readily bond with oxygen or sulfur under normal conditions. Pure gold holds its color indefinitely, though most jewelry uses gold alloys that include copper or silver, which reintroduce some tarnish risk.
Titanium and stainless steel also resist tarnish effectively. Stainless steel forms a passive chromium oxide layer that shields the underlying metal, while titanium stays nearly inert in most environments. Both are practical options for daily wear in humid conditions.
What speeds up tarnish in daily life in the UAE
The UAE’s environment creates unusually aggressive conditions for jewelry. Heat, humidity, coastal air, and daily product use all push the chemistry of tarnish forward faster than in cooler, drier climates. Knowing these specific triggers helps you understand why does jewelry tarnish so quickly for many residents here, and which daily habits to reconsider.
Climate and humidity
Dubai and Abu Dhabi sit near the coast, which means salt-laden air reaches your jewelry even when you’re nowhere near the water. Airborne sodium chloride deposits on metal surfaces and triggers electrolytic reactions that accelerate oxidation. Combined with temperatures that regularly exceed 40°C in summer, these conditions give moisture and oxygen exactly what they need to speed up surface reactions. Even air-conditioned indoor spaces contribute, since moving between extreme heat outside and cold interiors causes brief condensation to form on metal surfaces.
Salt air and rapid temperature changes are among the most aggressive tarnish accelerants a piece of jewelry can encounter.
Perfumes, lotions, and daily products
Fragrance plays a significant role in daily life across the UAE, and alcohol-based perfumes and oud oils both react with metal surfaces. The alcohol in sprayed perfumes creates a thin acidic film when it contacts jewelry directly, while sunscreen and moisturizing lotions leave residues that trap moisture against the metal. Applying any of these products before putting on a bracelet or ring speeds up surface oxidation noticeably. Take your jewelry off before applying any skin product, then let your skin dry completely before putting the pieces back on.
How to prevent jewelry from tarnishing
Prevention is more effective than restoration. Once you understand why does jewelry tarnish, most of the practical fixes come down to controlling your piece’s exposure to the three main triggers: oxygen, moisture, and sulfur compounds. Small adjustments to how you store and wear your jewelry make a significant difference over time.
Store it correctly
Where you keep your jewelry when you’re not wearing it has a direct impact on how fast it tarnishes. Airtight containers or anti-tarnish pouches limit the amount of oxygen and humidity that reaches the metal surface. For pyrite and silver pieces especially, place a silica gel packet inside the storage container to absorb ambient moisture. Avoid storing jewelry in bathrooms, where steam from showers raises humidity levels consistently.
Keeping each piece in its own sealed pouch also prevents metal-on-metal contact, which can scratch softer surfaces and accelerate surface wear.
Build protective habits
Your daily routine has more impact on tarnish speed than most people realize. Put jewelry on last, after applying perfume, sunscreen, and lotion, so chemicals don’t sit directly against the metal. Take your pieces off before swimming, exercising, or washing your hands, since chlorine, saltwater, and soap residues all accelerate surface oxidation. In the UAE’s climate, even a short outdoor session in humid summer air can leave enough moisture on a metal surface to start the process.
Wiping your jewelry with a soft, dry cloth after each wear removes sweat, oils, and airborne deposits before they have time to react with the surface. This single habit extends the appearance of any piece noticeably.
How to clean tarnished jewelry without damage
Once tarnish sets in, the right cleaning method depends entirely on what the piece is made from. Aggressive cleaning can strip surface finishes, scratch softer metals, or in the case of pyrite, introduce moisture that worsens the oxidation you’re trying to remove. The answer to why does jewelry tarnish explains exactly why harsh scrubbing or prolonged soaking isn’t the right approach: you’re dealing with a thin chemical compound on the surface, not embedded dirt that needs force to lift.
Gentle cleaning for silver and copper
Silver and copper pieces respond well to mild dish soap diluted in warm water. Use a soft-bristled toothbrush to work the solution gently over the surface, then rinse thoroughly and dry completely with a lint-free cloth. For heavier tarnish on silver, a paste made from baking soda and water applied with a soft cloth removes surface sulfide without damaging the underlying metal. Always rinse residue off completely, since moisture left sitting on the surface restarts the oxidation cycle immediately.
Drying your piece completely after any wet cleaning is as important as the cleaning method itself.
Cleaning pyrite safely
Pyrite requires a dry or near-dry approach because water accelerates iron oxidation directly. Use a soft dry cloth or a very lightly dampened cloth to wipe the surface gently, then dry the piece immediately. Avoid soaking, ultrasonic cleaners, and acidic solutions entirely. A careful polish with a microfiber cloth restores surface luster without introducing the moisture that triggers further tarnish on iron sulfide minerals like pyrite.
Key Takeaways
Tarnish is a chemical reaction, not a flaw in your jewelry. Oxygen, sulfur, and moisture bond with metal surfaces to form a thin compound layer that dulls the original finish. Understanding why does jewelry tarnish puts you in control: store pieces in airtight containers, wipe them down after each wear, and apply perfumes or lotions before putting your jewelry on, not after.
The UAE’s coastal humidity and heat make these habits more urgent than in most other climates. Whether you wear silver, copper, brass, or a mineral like pyrite, the environment actively works against your jewelry’s appearance. Small, consistent care routines remain the most effective defense against tarnish over time.
If you want pieces built for daily wear under these conditions, explore the handcrafted pyrite bracelets and accessories at Natural Pyrite UAE, where authentic materials and artisan craftsmanship are matched directly to the demands of life in the Emirates.



