Every pyrite piece we source and handcraft at Natural Pyrite UAE starts with the same fundamental chemistry: the pyrite chemical formula FeS2, known formally as iron disulfide. Two elements, iron and sulfur, bonded in a precise crystalline structure that produces pyrite’s signature metallic luster and remarkable hardness. That’s it. No coatings, no synthetic additives. Just nature’s own architecture.
Understanding this formula matters more than you might expect. It explains why authentic pyrite has a dark gray, steel-toned appearance rather than the bright gold finish you see on imitations flooding the market. It also reveals why this mineral earned the nickname "fool’s gold", and why the real thing, unpolished and unaltered, carries a weight and presence that no replica can match.
This article breaks down everything behind FeS2: its molecular composition, crystal structure, physical properties, and the science that makes natural pyrite a material worth knowing, whether you’re a collector, a geology enthusiast, or someone selecting an authentic pyrite piece for a Dubai executive suite or luxury residence.
What FeS2 means for pyrite
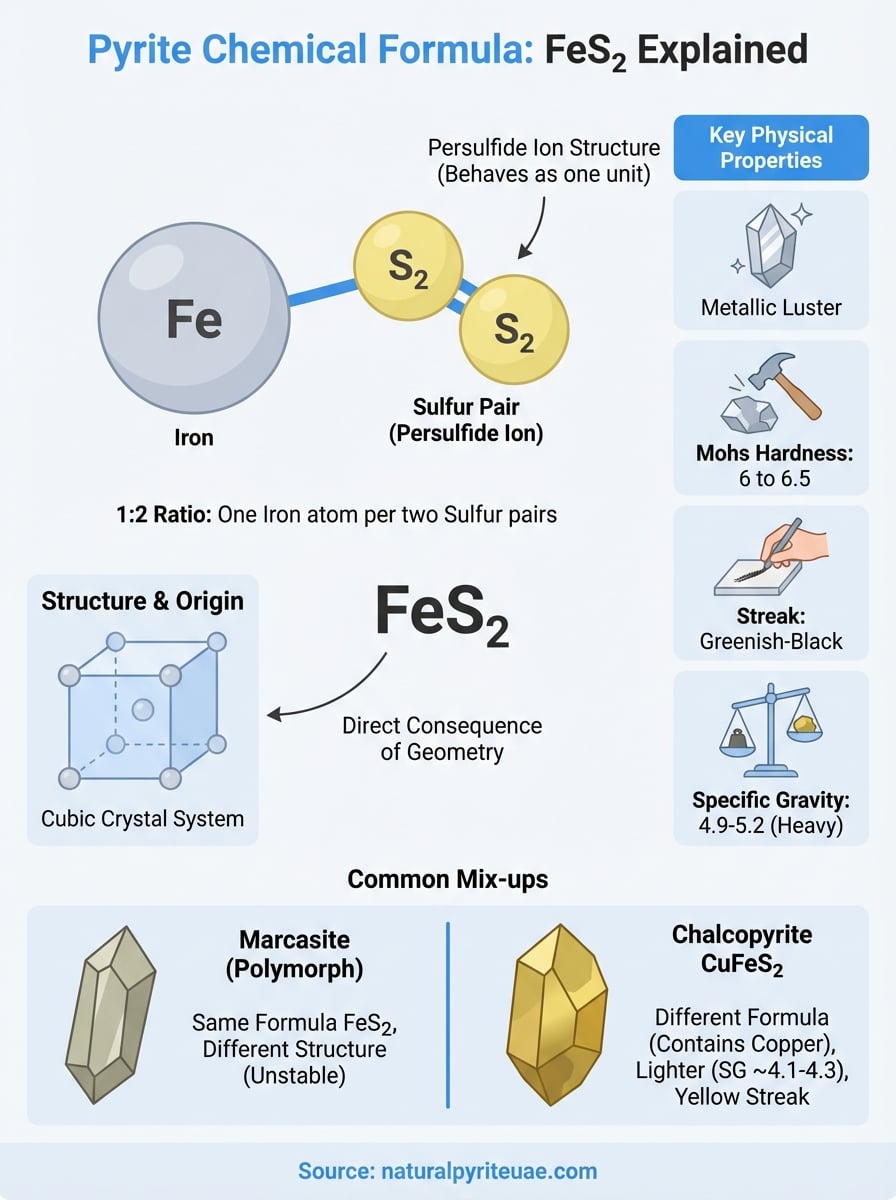
The formula FeS2 packs a lot of information into three characters. Fe is the chemical symbol for iron, derived from the Latin ferrum, and S2 tells you there are two sulfur atoms bonded to every single iron atom. That 1:2 ratio is not arbitrary. It defines pyrite’s entire character, from its density and hardness to the metallic shine that catches your eye across a room.
Breaking down the atomic ratio
Each iron atom in pyrite pairs with two sulfur atoms, and those sulfur atoms don’t simply sit beside the iron. They form S2 pairs, called persulfide ions, where each pair behaves as a single bonded unit. This arrangement is what separates pyrite from other iron sulfide minerals. The sulfur pairing locks the structure into place, giving pyrite a Mohs hardness of 6 to 6.5, which is harder than most common minerals you’d encounter.
The persulfide ion structure is the core reason pyrite behaves so differently from simpler iron sulfide compounds like pyrrhotite.
What this means for identifying authentic pyrite
When you know the pyrite chemical formula, you have a built-in reference point for spotting fakes. Genuine FeS2 produces a greenish-black streak when scratched across an unglazed ceramic tile, a test that imitations made from painted stone or resin will fail immediately. The formula also predicts a specific gravity between 4.9 and 5.2, meaning a real pyrite specimen should feel noticeably heavy for its size. If a piece feels light or leaves a yellow streak, the chemistry is off, and you’re not holding natural pyrite.

Why pyrite is called iron disulfide
The name "iron disulfide" follows standard chemical naming conventions used in mineralogy and chemistry. "Iron" refers to Fe, the element that forms the mineral’s backbone, and "disulfide" signals two sulfur atoms bonded to that iron. The prefix "di" simply means two, so the name maps directly onto the pyrite chemical formula FeS2. When you see this name in a geological report or on a product description, you can read it as an exact molecular statement about what the mineral contains.
Where the naming convention comes from
Chemists name compounds by identifying the elements present and indicating their quantities with Greek prefixes. One sulfur atom gives you a monosulfide; two gives you a disulfide. This system makes iron disulfide a precise label that tells any scientist the exact atomic ratio before they even examine the mineral.
The "di" prefix is your quickest confirmation that pyrite carries two sulfur atoms per iron atom, separating it chemically from minerals like pyrrhotite (FeS).
This naming approach also explains why pyrite and marcasite share the same chemical name despite being different minerals. Both contain FeS2, but their internal crystal arrangements differ, making them polymorphs. The name captures the chemistry alone.
How pyrite gets its formula from its structure
Pyrite’s formula doesn’t come from arbitrary chemistry. The FeS2 ratio emerges directly from how iron and sulfur atoms arrange themselves inside the mineral’s crystal lattice, making the pyrite chemical formula a structural fact as much as a chemical one.
The cubic crystal system
Pyrite crystallizes in the isometric (cubic) crystal system, which means its atoms stack in a highly symmetrical, three-dimensional grid. Each iron atom sits at the center of an octahedral arrangement, surrounded by six sulfur pairs positioned at precise, repeating angles. This geometry forces the 1:2 ratio of iron to sulfur, locking FeS2 into the formula permanently.

The cubic structure is also what produces pyrite’s characteristic cube-shaped crystals and the perfectly flat, mirror-like faces you see on high-quality specimens.
Because the sulfur pairs bond at fixed positions within this grid, no alternative ratio is stable. You cannot have pyrite with a different iron-to-sulfur count and still maintain the same crystal arrangement. That constraint makes the formula a direct consequence of geometry, not just a chemical observation, and it explains why every authentic pyrite specimen you handle carries the exact same atomic composition regardless of where it was mined.
Key chemical and mineral facts to know
Knowing the pyrite chemical formula gives you a foundation, but a few additional data points help you work with this mineral more precisely. Whether you’re evaluating a raw specimen or selecting a finished piece for a collection, these figures tell you exactly what you’re dealing with at a material level.
The molecular weight of FeS2 sits at approximately 119.98 g/mol, a number that reflects the combined atomic masses of one iron atom and two sulfur atoms.
Core mineral data at a glance
The table below summarizes the most important chemical and physical properties of pyrite in a single quick reference.
| Property | Value |
|---|---|
| Chemical formula | FeS2 |
| Molecular weight | ~119.98 g/mol |
| Crystal system | Isometric (cubic) |
| Mohs hardness | 6 to 6.5 |
| Specific gravity | 4.9 to 5.2 |
| Streak color | Greenish-black |
| Luster | Metallic |
These numbers aren’t just academic. When you handle a genuine pyrite specimen, the high specific gravity and metallic luster should be immediately noticeable. A piece that feels too light or shows a yellow streak is giving you direct chemical feedback that the mineral composition doesn’t match FeS2.
Common mix-ups with similar minerals
Two minerals get confused with pyrite more than any others: marcasite and chalcopyrite. Knowing the pyrite chemical formula helps you separate all three quickly, because the chemistry tells a different story for each one.
Marcasite: same formula, different structure
Marcasite shares the exact formula FeS2 with pyrite, which puts them in the same chemical category but not the same mineral class. The key difference is crystal structure. Marcasite forms in an orthorhombic system instead of pyrite’s cubic one, making it chemically identical but structurally distinct. Mineralogists call this relationship polymorphism.
That structural difference has real consequences: marcasite is significantly less stable and tends to break down over time, while pyrite holds its form for decades.
This instability makes marcasite a poor choice for decorative or collectible pieces. If you’re investing in a long-term display piece, pyrite’s cubic lattice gives it a durability that marcasite simply cannot match.
Chalcopyrite: a third element changes everything
Chalcopyrite carries the formula CuFeS2, introducing copper alongside iron and sulfur. That extra copper pulls its color toward a brighter, brashy gold tone, which is why people sometimes mistake it for pyrite. You can separate the two using a streak test: chalcopyrite’s specific gravity runs lower, around 4.1 to 4.3, so it will feel lighter in your hand than a genuine pyrite specimen of equal size.
Final takeaway
The pyrite chemical formula FeS2 tells you everything you need to know about this mineral before you ever pick up a specimen. One iron atom bonded to two sulfur pairs produces a cubic crystal structure that gives pyrite its metallic luster, high density, and long-term stability. That same formula separates it from marcasite, which shares the chemistry but not the durability, and from chalcopyrite, which adds copper and produces a brighter, less authentic appearance.
Understanding the formula also gives you a practical edge. You can use streak tests, specific gravity, and crystal form to verify what you’re holding, whether that’s a raw specimen or a finished piece. Real pyrite feels heavy, leaves a greenish-black streak, and holds its form for decades without degrading.
If you’re ready to bring authenticated natural pyrite into your space or daily wear, explore the handcrafted pyrite collection available in the UAE and find a piece built on genuine FeS2.



